The Hydrino Hypothesis Chapter 6
An Introduction To The Grand Unified Theory Of Classical Physics
This monograph is an introduction to Randell L. Mills’ Grand Unified Theory of Classical Physics, Hydrino science, and the efforts of the company Brilliant Light Power (BLP) to commercialize Hydrino-based power technology, as told by Professor Jonathan Phillips. Out of necessity, it assumes a degree of familiarity with physics and physics history. An overview of the BLP story which serves as a helpful introductory piece to those unfamiliar with its sweeping scope can be found here. Readers should also read the previous chapters of this monograph prior to this one:
Chapter 1 of The Hydrino Hypothesis
Chapter 2 of The Hydrino Hypothesis
Chapter 3 of The Hydrino Hypothesis
Chapter 4 of The Hydrino Hypothesis
Chapter 5 of The Hydrino Hypothesis
By Professor Jonathan Phillips
Experimental Verification Against Accepted Data for Hydrogen Excited States
Preface- In this chapter it is shown that the observed spectra arising from electrons in excited states of hydrogen are predicted by the GUTCP; however, excited state predictions require a new understanding of the nature of photons and their impact on the forces between particles. An overview of the GUTCP conception of photons and how they interact with electrons is provided below.
Introduction
The next success of the GUTCP in predicting the behavior of established forms of matter is the near-perfect agreement of the predictions of the theory with the repeatedly measured energy levels of the excited states of hydrogen.
To put it another way: the GUTCP predicts, with no variable parameters, all the known, thoroughly documented, excited state energy levels of hydrogen. This agreement with data is set as a distinct chapter as it represents the first example of a modification of a classic physics law required by the GUTCP.
Any hypothesis that involves a modification of classical physics laws requires intense inspection. Moreover, the Hydrino Hypothesis, the subject of the next chapter, requires some understanding of the nature of transitions between energy states and photons according to the GUTCP. In this chapter the GUTCP photon is introduced.
It is also important to note that the GUTCP model of excited states includes some novel concepts of the nature of the photon. The mathematics is still classical physics, and the forces are still those of classical physics, but photons are re-imagined.
Specifically, the photon as it pertains to excited states:
Is an element of electromagnetic energy trapped in an orbitsphere resonant cavity, and;
This trapped photon modifies the force existing between the proton and electron in hydrogen (and other atomic species).
Moreover, these concepts will be shown, in the next chapter, to be key components of the model of hydrogen states below the conventionally accepted ground state.
That is, these concepts are at the heart of the Hydrino Hypothesis.
Thus, it is important that these concepts be discussed in some detail not simply to show another example of the success of the GUTCP with Other People’s Data (OPD), but also for understanding the process of Hydrino formation.
These key concepts, which are incorporated into the Newtonian/Maxwellian force balance equation, per this author’s best understanding of the Source Text, are:
A: A photon is required to (temporarily) create excited states of orbitsphere electrons. Mathematically, photons are described as trapped radiative energy of specific allowed values, in spherical resonant cavities of very specific allowed sizes. In an excited state, a photon is “pasted” to the electron current loops which compose the orbitsphere.
B: Only an electron + photon (an excited state) of specific energy and size can form, because both the force balance and known properties of excited states must be simultaneously satisfied.
C: The photon has a radial electric field that reduces the net force existing between the electron and the proton(s) in the nucleus. Thus, the force balance equation for the electron is modified in a very specific fashion by the trapped photon.
D: Notably, the photon has no actual charge, hence the local presence of a photon field, which net reduces the effect of the proton field in the force balance of the electron, does not change the net neutrality of an atom.
E: An electron/trapped photon entity is inherently unstable because the photon standing wave has components congruent with the speed of light. It has been repeatedly demonstrated using the classical physics model of electromagnetism that any object with components congruent with the speed of light is unstable and thus radiative.
Before proceeding it is absolutely incumbent on this author to note that the discussion of the energy levels of excited states is the only topic regarding excited states of hydrogen that is considered herein in any depth.
In the Source Text, this is only an introductory subject.
Other topics covered in the Source Text include:
Instability of excited states, very briefly reviewed herein.
Source currents of excited states.
Selection rules.
Orbital and spin splitting.
Stark effect.
State lifetimes and intensities.
Resonant line shapes.
Hydrogen Lamb shift.
Hydrogen fine structure, and much more.
The Source Text discussion of excited states is a tour de force, like a set of PhD theses of the highest caliber. Thus, this chapter is only intended to be a brief introduction.
In any event, concepts A-E above have no parallel in SQM and do not belong to any of the canons of standard physics. In standard physics the photon is difficult to track, as attested by the famous physicist Richard Feynman in a conversation he recalled having with his father:
“I understand that they say that light is emitted from an atom when it goes from one state to another, from an excited state to a state of lower energy.”
I said, “That’s right.”
“And light is kind of a particle, a photon, I think they call it.”
“Yes.”
“So if the photon comes out of the atom when it goes from the excited to the lower state, the photon must have been in the atom in the excited state.”
I said, “Well no.”
He said, “Well, how do you look at it so you can think of a particle photon coming out without it having being there in the excited state?”
I thought a few minutes, and I said, “I’m sorry; I don’t know. I can’t explain it to you.”
-Richard P. Feynman, The Physics Teacher (September 1969).
The standard SQM physics paradigm, for photons, per above, is clearly very “Alice in Wonderland.”
In contrast, per the GUTCP, the photon is described as a trapped form of electromagnetic energy which is materially present in atoms in excited states. Upon decay of the excited state, the photon is released, again a material object, as electromagnetic energy (e.g. light). It never disappears and it is always possible to account for the energy associated with it.
As discussed previously, photons released from excited state hydrogen (and all other atomic species) are emitted at very specific frequencies. Figure 6-1 below illustrates these discrete series of photons that correspond to specific excited states.
Figure 6-1: Excited states and transitions to lower energy states of atomic hydrogen. Prominent spectroscopic series labelled. Notably only the Balmer series is in the visible range, hence Balmer lines were the focus of early studies of quantized energy levels.
Key Concepts
Below, each of the above key concepts is discussed in more detail. Very little mathematics is presented in the discussion below because the mathematics of resonance can get complicated quickly, and detailed mathematical development is available in the Source Text.
Concept A: Resonators
Resonators are common in our world. For illustration, a couple of macroscopic examples of the sort familiar to any observant individual are reviewed. Moreover, the use of such macroscopic analogs is totally legitimate, as the GUTCP is based on the requirement that physics is the same at all scales, from the cosmos to the quantum level. There is no problem postulating physical resonators of the atomic scale in the GUTCP.
The first example is a guitar string. Before the string is picked two boundary conditions are already in place. Specifically, the string is held in place at two locations:
A finger holding it down on the fingerboard.
A pin holding the string on the bridge.
These points, in the language of physics, are boundary conditions. The string does not move at these points, whereas at all other points, the string is permitted to move.
Next, the string is “picked.” The actual pick that starts the movement of the string is, in the mathematics of physics, a wave packet. In classical physics any disturbance that is not infinite in extent, physical or electromagnetic, can only be described as a weighted mixture of waves of all frequencies.
The string is picked, that is, physically moved/disturbed, over a small area for a very short time. The physical pick is mathematically described as a wave packet because it is not infinite in time or extent. Thus, the motion of the string after the pick is described as containing many frequencies, although the practical range of vibrations of significant magnitude actually occupies a small region of frequencies.
That is, only a relatively small number of frequencies have significant intensity. In general the wave packet intensity is highest at a mean frequency, and the intensity of frequencies away from this average drops quickly.
The next process is one of filtering. The system filters out most of the frequencies of the pick. Only those frequencies that match the boundary conditions of the string, as described above, will not quickly disappear, or in the language of mechanics, those frequencies that don’t match the boundary conditions are damped out.
What are those non-damped, or “resonant,” frequencies that are not filtered? They are frequencies that match the length of the string, such that at the boundary points at the end no motion takes place. There is a longest wave, represented by motion that moves the string over the entire length with maximum displacement at half length, and then overtones of shorter wavelengths in which the standing wave has one or more zero-motion points between the endpoints.
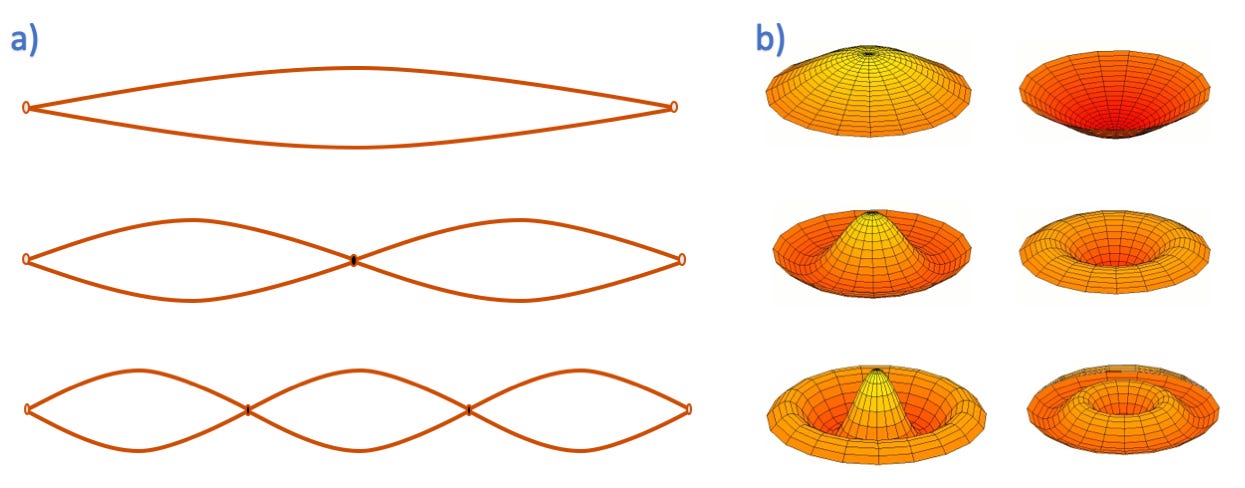
Indeed, high speed photography actually shows standing waves with zero motion points on picked strings. The collection of permitted standing waves is illustrated in Figure 6-2 (a).
Figure 6-2 (a): Standing waves in two dimensions. Permitted standing waves lengths on a string held at two ends. Figure 6-2 (b): Standing waves, several harmonics on a circular membrane.
The wavelength is not the same as the sound frequency. There are many contributions to the frequency, hence sound, of the vibration. Frequency and wavelength are related, generally by a simple equation with several parameters which are determined by the physical nature of the string.
Clearly, the heaviest/thickest string of a guitar and the lightest string can have the same length standing waves yet very different vibration frequencies.
A second illustration is a drumhead, as seen in Figure 6-2 (b). The vibrations on a drumhead membrane are similar to that of a string in that only vibrations that match the forced boundary conditions (the zero motion position created at the intersection of the membrane and the drum body) of the drum are possible.
Similar to the frequency of the vibrations on a string, the wave shapes are related to the frequency of the vibration, but that relationship is a function of the nature of the membrane material, the tension (tightness) of the membrane, etc. Waves that match the fixed boundary conditions of the two-dimensional drumhead are also called standing waves as they do not change shape with time, but rather simply stand in place.
The drumhead model is particularly germane to the concept of the GUTCP’s electron orbitsphere, as the orbitsphere is also a membrane with boundary conditions. In particular, the orbitsphere boundary conditions are simply that any allowed standing wave must be periodic. That is, it must have the same value at zero and 2 pi radians.
Happily, for spherical membranes, the mathematics of allowed standing waves on a spherical surface was worked out long ago. The allowed behavior is described by the angular solutions to the Laplace equation, with all other vibrations damped out. In brief, only waves that are described by the angular solutions to the Laplace equation are stable on orbitspheres. This implies that not only does a vibration with a single node exist (similar to Fig 6-1), but a set of overtone frequencies as well. As noted in the Source Text, the radial term (separation of variables) solution is a delta function, that is, the standing waves form on a “standard” orbitsphere of constant radius.
A little too much detail: these overtone frequencies give rise to other states, known in the SQM model as the “p,” “d,” and “f” electron states. All of the known spectroscopic states (p,d,f) are predicted to exist in the GUTCP.
The mathematics of Laplace Equation angular solutions are such that there is a principle number “n.” The overtone states, the “l” quantum number, cannot stabilize unless n=2 for the “p” states, n=3 for the “d” states, etc. Significant to this discussion is the fact that only the “n” number is important for determining the energy level of allowed excited energy states in hydrogen.
In the absence of more than one electron, all the l-state energies are “degenerate,” that is energetically identical, hence spectroscopically indistinguishable. So, for hydrogen, only the n value is known (Figure 6-1). For all other atoms coupling affects the energy and the l-states for a particular “n” are not energetically degenerate. (In the case of multiple electrons the l-state energy levels are not degenerate per the GUTCP Mod 1 model, but further discussion is beyond the scope of this monograph.)
We’ll stop this discussion here. The intention here is only to provide an intuitive introduction to “resonance,” not a full-bore explanation.
B: Quantized Size
Photons that create a Laplace Equation angular solution type excitation of the orbitsphere, and only such photons, can be trapped. Moreover, according to the physical model of the GUTCP, the trapped photon in the resonator cavity of the orbitsphere is essentially pasted to the orbitsphere.
The allowed standing waves on a spherical surface, with and without a trapped photon, correspond to the allowed quantized states in the GUTCP.
Moreover; mathematically they have a one-to-one correspondence with the “s,” “p,” and “d” states of SQM. For example, just as there can be no quantized spherical harmonics for “s” states (n=1) in SQM, there can be no quantized harmonics for the “s” states in the GUTCP. Just as there are harmonics for n=2 in SQM, there are the same number of Laplace Equation angular solution states for n=2, n=3 etc. in the GUTCP.
So, every known quantized state in atomic spectroscopy is predicted to exist in the GUTCP.
C: Force Balance of Excited States
The actual determination of the allowed size of electron/photons resonant cavity is, just as for the ground state, determined, per the Source Text, by a force balance:
In this force balance, the interaction strength of the force between electron and proton is inversely proportional to n, the principle quantum number (Figure 6-1).
For the ground state, this value is one, and the equation reduces to that discussed earlier (Ch. 4). It is asserted in the GUTCP that the force strength reduction expresses the net effect of a photon. In this theory, the photon creates a local field oppositely polarized to that of the proton, thus reducing the force exerted by the proton on the electron of hydrogen, yielding an excited state.
The 1/n relationship of Eq. 6-1 is not entirely classical. In classical physics, the field created by an object is generally not considered size-dependent. Yet in the GUTCP it is asserted the photon field is a function of n, hence radius, of the orbitsphere.
Physically, why would a resonant cavity photon in a larger size orbitsphere block the action of the proton field better than that of a smaller, lower n value, cavity?
There are two answers to this question:
The Source Text presents a complex explanation which concludes this relationship in the force balance must pertain for fundamental reasons. This explanation is beyond the scope of this article and the interested reader is encouraged to review the Source Text for full detail.
It is allowed, in fact it is necessary, in classical physics to postulate mathematical relationships in force and energy balances. If the mathematical model is successful in describing fundamental data, in making quantitative predictions, it is accepted until such time as it is shown to be incorrect. There are many examples of this postulation of mathematical force/energy relationships in the history of physics. The ones we are familiar with are those that passed muster by being in agreement with experimental evidence.
Historical Example I of a postulated relationship in a force balance: the original suggestion that the force of gravity of the sun acting on the planets is inversely proportional to the distance between them squared, 1/r2, was based on fitting experimental data.
The data might have instead suggested an empirical relationship of ~ 1/r 2.1 , etc, and the model would have changed to reflect this. This mathematical expression of gravity being proportional to 1/r2 is now generally considered a fundamental law of the cosmos. It is the “law” used to predict the orbit of planets, comets, guide spacecraft with great accuracy, and suggests there is some alien force at work when a cosmic object does not obey the rule.
As the relationship was not declared to mankind by a deity and is merely a well-tested empirical relationship, it can be, and in fact is, regularly challenged.
One significant recent challenge was presented by M. Milgrom and has led to the notion that the Newtonian version of gravity can be modified as an alternative to the suggestion that there is invisible/non-baryonic matter in the galaxy/universe.1234
The suggestion that gravity may not always have the inverse square behavior that is currently regarded as a “fact” is drawing increasing attention and experimental support.5
The particular issue of dark matter and its relation to the precise form of gravity will be discussed in greater detail in Ch. 10 of this monograph.
In brief, there is no basis for rejecting the modification of a term in an energy or force balance in classical physics if the modification is consistent with observations. Of course, if additional observation demonstrates the proposed new rule is incorrect, it must be abandoned. In the case of the resonator cavity treatment of the GUTCP, the behavior is ascribed to a structure not hitherto postulated, and as shown in Table 6-1 below, the predicted values match the observed values with spectacular accuracy.
At present the proposed very simple force balance relationship passes all requirements of classical physics.
Historical Example II of a postulated relationship in an established balance equation: Standard Quantum Mechanics. As noted in earlier chapters, the Schrodinger equation is essentially an energy balance and it is modified from the classical energy balance in many ways.
It requires that no macroscopic rules of classical physics pertain at the quantum level. SQM completely dismisses the application of Newtonian trajectory, for example. Despite this explicit repudiation of classical physics, SQM was worthy of consideration because it matched the empirically determined spectra of hydrogen. It fails, however, as covered in great detail in Chapters 4 and 5, to explain any atom with more than one electron, even the relatively simple helium atom.
Other examples of empirically determined properties worth considering: the expression for electric field strength, the permittivity of free space, Planck’s constant, the mass of the electron, the Special Theory of Relativity, etc. It is interesting to note that in the GUTCP there are predicted values of several of these empirically determined constants: the mass of an electron and Plank’s constant.
The agreement of the measured values of the mass of an electron and Plank’s constant with the no variable parameter predictions of the GUTCP are further demonstrations of the enormous power of the GUTCP. This subject, however, is beyond the scope of the present monograph.
D: No Charge
A second major property of photons required to match experimental observation is that they have no field. If photons had fields equivalent to a particle (e.g. an electron), then an excited state hydrogen atom would be charged. That is, the charge on the electron orbitsphere, with no trapped photon, balances the charge on the proton in a ground state hydrogen atom. The net observable charge outside the orbitsphere is zero. The fields cancel.6
If a photon added charge to the atom, there would be a net charge on any hydrogen atom in an excited state. Electron and proton fields would still cancel, but nothing would be available to cancel the field created by the charge of the photon! Ergo, for the GUTCP to match observation, there must be no charge on a photon. This should not be a problem because photons, or any form of electromagnetic radiation, are known empirically to be without charge.
In sum, per points A-D, the GUTCP model predicts energy levels for excited states of hydrogen. This is done using simple algebra based on classical physics. Still, the classical physics force balance is modified in a simple manner as a function of the principal quantum number n.
It is argued that there is no prohibition against modification of a proposed force relationship in physics, hence proposing a modification is a permitted activity, as was done with the requirements of SQM. The only judgement regarding the validity of the proposed change is not the judgement of experts, but rather is based on experimental data. That is, validation of the proposed force balance modification of the GUTCP only requires that the proposed modification of the classical equation matches experimental data. In this case it clearly does.
In the next section, an explanation for the instability of the excited states and the stability of the ground state are given.
E: Stability
In the GUTCP it is claimed that by classical rules of electromagnetism, some orbitspheres should not radiate. This has been demonstrated mathematically, as well as experimentally, for macroscopic systems. The classical physics, (no SQM), derivation of the non-radiative behavior of circulating current loops of constant angular momentum was developed by several physicists.789 The mathematical developments are quite complex and will not be reviewed here.
As noted, the mathematics leads to an exception to the rule of stability of continuous current loops. In particular, the exception to the rule is that any circulating current that contains components synchronous with the speed of light will not be stable and will radiate. As discussed more below, an orbitsphere electron with a photon pasted to it contains components synchronous with the speed of light and hence is not stable.
A physical explanation of the concept of non-radiation is attempted here:
Any current moving slower than the speed of light that does not disturb the existing field structure of the elementary particles that make up the current will not radiate.
A particle moving at constant velocity will not radiate. A loop of steady current will have a steady state field and will not radiate.
Why? At every instant in time, at any point in space, the arrangement of charges that create the field is unchanged. The charge distribution is constant in time and hence will produce an unchanging field at every point in space.
In contrast, the field of a small particle traveling the same loop path varies in time. Example: the field at a Bohr “planetary” point electron’s orbit of the nucleus will be a function of the particle’s position on the loop. Assume the observer is to the right of the Bohr atom. The field due to the orbiting electron will be weaker when the particle is on the “left” side and relatively stronger when the particle on the right side, hence closer. At every point in space the field will be changing in synchrony with the moving planetary electron. A change in the field requires energy, hence radiative energy loss by the particle traveling the loop.
In the GUTCP Source Text it is demonstrated that the combination of an electron orbitsphere with pasted photon in excited state hydrogen always has (photon) components synchronous with the speed of light.
For this reason, no excited states in hydrogen are stable.
They are all expected in the GUTCP framework to radiate a photon and enter a lower energy state. In contrast, the classical ground state of the hydrogen electron in the hydrogen atom (-13.6 eV) has no trapped photon according to the GUTCP model.
In the absence of a photon, the orbitsphere is stable because:
There are no components synchronous with the speed of light.
The great circles of current that compose the orbitsphere are loops of charge, and as noted, in classical physics steady loops of current do not radiate.
Hence, if the hydrogen electron is initially in an excited state it will quickly collapse to the first (First? See next chapter.) stable state, that is the standard ground state of -13.6 eV.
As noted, data derived from experiments is the ultimate arbiter of validity. Theory is interesting, but facts rule. Is there experimental evidence that loops of charge will not radiate?
Absolutely!
It has been demonstrated with loops of current in superconductive material. To quote one of the earliest reports on persistent currents in hard superconductors (HSC):10
“The fastest decay rate yet observed is dH/dlog t = 10 gauss per decade, from an intial state of M = 1000 gauss. If this rate of decay continues indefinitely, we estimate that the persistent current in this HSC sample will die out after 3X1092 years. In any practical sense then, the persistent current is persistent.
It is interesting to read the standard quantum model of persistent currents in superconducting materials, particularly the model promoted by the infallible (Not!) Wikipedia.11
In sum, the GUTCP model of the excited states of hydrogen perfectly predicts the energies of those states. The model requires that excited states consist of electrons in an orbitsphere configuration and photons resonantly captured by the orbitspheres.
The trapped photon reduces the force between electron and positive charge in the atomic nucleus. In turn, the classic theory of persistent current indicates excited hydrogen states should be unstable, quickly losing their photon and moving to the photon-free traditional hydrogen “ground” state, as observed. The “ground” state is stable as it contains no photons, hence no frequency components at the speed of light.
In sum, the GUTCP passes another test: it predicts the energies of the known excited states of hydrogen, and the instability of excited states, all on the basis of classical physics. As shown in the Source Text, it also is in quantitative agreement, with no adjustable parameters, with properties such as the Lamb Shift, relative intensities, Stark Effect, and other known properties of hydrogen. As will see in future chapters, it also predicts a new state of hydrogen: Hydrino states.
Personal Note
Future iconoclasts, prepare to be uncomfortable. The mildest discomfort one will need to embrace is the absence of professional accolades. You will not be invited to be the Keynote Speaker at any conference and your papers will not garner enormous numbers of citations, if any. You will not edit journals.
You will also need to embrace the Lone Ranger approach to professional life. Indeed, the hiring process was, is, and forever will be organized against the iconoclast. Schools hire faculty from the research teams of faculty members from leading departments, generally defenders of the standard paradigm.
The physics departments at the “leading schools” will never consider hiring a Randy Mills. And schools lower on the Totem of Academia are even less likely to hire an iconoclast.
As such, the iconoclast will need to find support outside the standard government grant/professor route to perform research; difficult, but as demonstrated by Randy, not impossible. I joke (?) that Randy’s real sin in the eyes of the scientific community was not breaking the quantum paradigm, but rather the government grant funding paradigm.
The situation is not entirely hopeless. One of the great influences in my life was Bob Bird (R. Byron Bird), the lead developer of what is now a key component of engineering education: Transport Phenomenon, a course, a book, and a foundational conceptional structure devised by Bob and two collaborators in the late 1950s.
Variations on this theme are now found in virtually all modern engineering curriculum. Surprisingly (?), in hindsight, the adoption was not instantaneous. Bob was hired by Cornell University and then U. Wisconsin-Madison, not because of this book, but rather because of his impressive work with his advisor, Joe Hirschfelder, a recognized leader in physical chemistry.
Still, despite holding faculty positions in chemical engineering, Bob told me that he and colleagues were forced to teach the course, not in Chemical Engineering, but in Nuclear Engineering for the first couple of years. He explained: chemical engineering was already an old discipline, its core tenets jealously guarded by senior faculty across the country. Whippersnappers are, and were, dis-invited from making efforts to change the discipline’s structure!
The negative reaction to the new proposed course in Transport Phenomenon was not merely a local phenomenon at the UW-Madison, but at the national level as well. Bob, after some episodes of rejection, held a rather low opinion of the American Institute of Chemical Engineering for his entire professional life.
In contrast to chemical engineering, nuclear engineering was in the late 50’s and early 60’s a new, entrepreneurial field, with denizens eager to explore new ideas, and develop a unique intellectual core. The culture of the moment in nuclear engineering was to seek new ideas, not find old experts.
An analogy can be made to the story of IBM vs. Apple. IBM, a behemoth of its time, could not break free of the constraints of its own paradigms, such as the supposed superiority of large computers servicing many individuals, hence; Apple, Microsoft and other tiny companies led by entrepreneurial twenty-somethings, had free rein to develop the first personal computers, and the accompanying software. The mid-story is a bit complicated, but we know the final chapter.
Indeed, younger readers will probably think: Apple is fantastic. Love Microsoft! But they may very well ask: what is IBM? I’ve got my Tesla in the garage. What’s a Chrysler?
In any event, Transport Phenomenon is now the core of the core of the chemical engineering curriculum in virtually every program. Perhaps, like IBM, chemical engineering would have faded into irrelevance if not for the effort of Bob and colleagues.
And Uncle Bob went on to become a member of the National Academy of Engineering and National Academy of Science, and won the National Medal of Science and a multitude of other honors. He had so many awards and plaques I assumed he needed a big closet to hold them! They were not displayed.
My own story of meeting Bob Bird is amusing. It’s a long story which begins thus: I came to be an honorary member of the Chemical Engineering Dept. at U-Wisconsin via a most unusual route.
First, I decided to attend graduate school after graduation from my small liberal arts college in Vermont because no career was interesting or beckoning. And the Vietnam war, or more exactly my opposition to it, had generated in me a jaundiced perspective on the establishment. In short, I was not eager to join it.
Next, I only applied to a few schools because there was no online then. Each application was a bona fide, independent, unique hard copy process, and I am a poor typist. Poor typists can be successful with Word, etc. because it is so easy to correct errors, but what today’s whippersnappers don’t know: it was not so easy on a real typewriter! Using a computer to help fill out forms was, at best, complicated at the time.
Second, my grades were merely very good (dedicated hippy and cross-country skier), not excellent, but my GRE scores were excellent. That apparent contradiction certainly requires multiple applications, as each program regarded such mixed messages differently. So, I came up with an appropriate plan: I applied to a stretch school, which was my first choice possibly because of its strong anti-war reputation (UW Madison), and some safe schools (Colorado and Maryland).
In the end, I was only accepted at my preferred school, and they wanted me in the new Materials Science program rather than Physics. They offered me a full ride and support, and predicted I would graduate in five years! Physics programs of the time were notorious for providing spotty support and taking f-o-r-e-v-e-r to complete.
Final analysis: karma took a turn, and for the best.
At the time, materials science was a new idea, a new program looking to create a core curriculum. There were no trained material science faculty, as there had never been a material science discipline. The solution was to build the program using faculty members who were attached to a host of departments: metallurgy, physics, chemistry, chemical engineering, nuclear engineering, and others.
Each discipline had aspects that related to materials, hence the net for the program would be a true overview of materials. My scholarship was not tied to a particular faculty member or discipline. In fact, the program was designed to allow me to survey the field and select a direction during my first summer. Still, I was assigned, as a starter project to work on in summer ’76, to a materials program related to the design of a nuclear fusion reactor. (Note: My advisor, Dick Post, went on to be a founder of AsTex, the company that sold me several of the microwave systems I used for the Balmer line broadening studies which will be discussed in Ch. 8.) At the time, nuclear fusion was poised to reach commercialization in 30 years. I believe it is now only 40 years in the future.
It transpired that I was not spiritually in tune with the fusion program. I was one of a host of graduate students, each working on a small part of a big plan. My part was to study the impact of high radiation flux on the graphite wall proposed for fusion reactors. Others were working on laser systems to probe the temperature and composition of the plasmas in the fusion reactors, others were studying the interaction of lithium with radiation as the notion of the time was that liquid lithium would be used to capture the energy of particles generated by fusion, others were doing plasma modelling, etc. I felt like a micro-ant in a big anthill. I was looking for something that was really mine!
I “interviewed” possible advisors in physics, chemistry and even chemical engineering faculty regarding projects under offer. I selected a young (25!) professor in chemical engineering as my advisor as the project he offered sounded like advanced physics to me, yet on a scale appropriate for a single investigator.
To wit: use Mossbauer Spectroscopy to determine the morphology of nanometer-scale particles supported on high surface area supports. That is, the project core was the study of the exact nature of the iron-containing heterogeneous catalysts used in ammonia synthesis, water-gas shift production of hydrogen, etc.
I must confess I knew nothing of chemical engineering, not the area or work or study, not the culture, and certainly not the curriculum. Once committed to the project I needed to figure out the terrain.
It began with natural interactions with my chemical engineering peers. I discovered they, at least the four others on our catalysis team, projected a certain arrogance. I was surprised, having spent the prior 4 years embracing the culture of physics, which is an elitist culture.
Indeed, anyone marinated in physics culture believes the denizens of all other fields of science, particularly any field of chemistry, are lesser beings. Given my marination, I was stunned to find my engineering peers to have a physics-like arrogance. In fact, there were non-subtle indications they did not believe I had the right to belong to their elite club! How misinformed they must be, I felt.
Moreover, I had never met any real engineers prior, as my small college only had the science component of STEM education, and I grew up in the DC area/swamp where engineers are few and far between. These were the first engineers I ever encountered. Were all engineers arrogant?
After only about a month in my new research team, my question about engineering arrogance led to a peculiar meeting with my new advisor. I asked my advisor: “What is the ranking of the Chemical Engineering program here at U W-Madison?”
This question arose from the “arrogance” I was observing. Could I have stumbled into a special program? My advisor took a minute, studying me, looking perplexed. Finally, he answered: “We like to think we are Number One.” I believe he wondered if I was being disingenuous. In any event, I recall spending a minute being confused. Was he being disingenuous? And if his statement were true, why?
A few weeks later I was invited on a day hike led by a Professor Bird, who would also provide transport (Pun intended). We were to meet in front of the Department at 8:00 am Saturday. This was perfect for me as I was too poor (sub-minimum wage scholarship) to afford anything but cheap food and rent for my shared cockroach infested apartment. Car? Forget about it! Exploring the wilds of Wisconsin, something I very much wanted to do, was going to require hitching rides with navigators. Thus, the invitation was perfectly aligned with my plans.
The day-hike event consisted of climbing the Moraine Hills around Devil’s Lake, a beautiful Wisconsin State Park that I suggest y’all visit, about an hour drive northwest of Madison, WI. Long haired and hippy looking, I spent hours, up hill and down dale, discussing the involvement of the US in the Vietnam War with Prof. Bird.
I clearly, but politely, presented the anti-war argument, and Prof. Bird, closely cropped hair and proper, explained the need for the US to counter communism, etc. I completely respected the man. (Later I learned he had served in the front lines of WW II in Europe, crossing the famous Remagen Bridge the first day and surviving the blistering counter-attack by German forces. Bob Bird, my take: Hero.)
All seemed perfectly OK to me; however, when we returned to Madison, my two peers, quiet all day, told me I was “finished.” Why? Well, obviously because I had spent the better part of an entire day arguing with Prof. Bird!
My question: why is Prof. Bird so special? Well, they explained that the origins of Transport Phenomenon, that it was this that made the department special and No. 1 and accounted for the fact that virtually the only students to qualify for the graduate program were those that had graduated No. 1 from their undergraduate Chemical Engineering programs. And I had the temerity to argue with the great professor all day!
Who was I, who had obviously connived to get into this exceptional program via the back door of materials science, to argue with anyone? After all, they had studied and worked around the clock for years to attain the same end point. I interpreted the hiking event differently. From my perspective, Bird and I had not argued, just discussed with mutual respect. After all, he was my ride! Couldn’t afford to piss him off!
The days passed. Friday arrived. The word of my impending doom had been shared with all the grad students in the program, numbering more than 100. I was in a big lab, perhaps 20 meters by 10 meters, sharing the experimental space with at least six others when the door opened and I heard Prof. Bird ask: “Where is Jonathan Phillips?”
Suddenly there was silence in the room. We all considered that my doom had arrived. All the others simply pointed to the very farthest corner where I was working. Prof. Bird boomed: “Jonathan, I had such a great time last week. Will you join us for another hike tomorrow?”
This man, I realized, was great man. No cancel culture tendencies cluttered his mind. Karma found me a great friend with whom I shared many adventures.
One lesson I quickly learned is that engineering requires real-world tests of ideas. Ideas leading to rabbit holes are not endorsed. Models and concepts that solve real-world problems get traction and accolades. For this cultural reason the authors of Transport Phenomenon could not be ridiculed for too long. The use of the ideas therein were quickly put to use improving the performance of all sorts of “Unit Operations” in real chemical plants, refineries, etc. Hence, they remained in engineering purgatory for a period short by historical measures; perhaps only a decade.
This purgatory story for new ideas is repeated many times in history. Consider the Wright Brothers episode. Once real planes were built by the Wright Brothers and flown in front of spectators for more than an hour at Lemans, France, it was no longer possible to dismiss them as mere bike mechanics.
In contrast to engineering, in many branches of physics, there are no real tests. Indeed, there will be arguments about the nature of dark matter forever (Ch. 10). The origins of particular spectral lines coming from intergalactic matter will be a forever discussion. That is, unless Randy’s work is paid attention to (Ch. 8).
Politics, personality, and money can all play an outsized role in such an environment. Perhaps, weirdly, even where there are real tests, as per the many examples provided regarding the GUTCP in this monograph, the illogic of “there are no real and reliable tests” appears to prevail.
In some cases, some concepts (e.g. SQM) become belief-based, like religion. After all, the electron can never be directly probed!
Whatever.
Sometimes it is almost like belonging to a tribe: you cannot belong until you demonstrate acceptance of the doctrine of the high priests, e.g. genuflect to SQM. Religion, politics, cancel culture, bullying, are all part of the physics culture. And so much of the rest of academia.
The situation has not and will not change. Can the non-Woke be hired at any universities? Can they even be invited to present at seminars? Will they be shouted down? Perhaps there will be physics departments which will require SQM loyalty oaths of incoming students and faculty. Seriously. This monograph will certainly not lead this author to garner any invitations to speak at physics colloquia or get physics accolades.
M. Milgrom, “A modification of the Newtonian dynamics as a possible alternative to the hidden mass hypothesis.,” The Astrophysical Journal 270, 365–370 (1983).
M. Milgrom, “A modification of the Newtonian dynamics: implications for galaxies.,” The Astrophysical Journal 270, 371–383 (1983).
M. Milgrom, “A modification of the Newtonian dynamics: implications for galaxy systems.,” The Astrophysical Journal 270, 384–389 (1983).
K.-H. Chae, “Distinguishing Dark Matter, Modified Gravity, and Modified Inertia with the Inner and Outer Parts of Galactic Rotation Curves,” ApJ 941(1), 55 (2022).
J. Phillips, “Theoretical and experimental basis for the super dielectric model of dielectric materials,” Phys Essays 33(3), 306–318 (2020).
G.H. Goedecke, “Classically Radiationless Motions and Possible Implications for Quantum Theory,” Phys. Rev. 135(1B), B281–B288 (1964).
H.A. Haus, “On the radiation from point charges,” American Journal of Physics 54(12), 1126–1129 (1986).
T.A. Abbott, and D.J. Griffiths, “Acceleration without radiation,” American Journal of Physics 53(12), 1203–1211 (1985).
Y.B. Kim, C.F. Hempstead, and A.R. Strnad, “Critical Persistent Currents in Hard Superconductors,” Phys. Rev. Lett. 9(7), 306–309 (1962).








Is Randy having n investors meeting this April John Cusenza
Thank you very specifically for your personal note regarding your early years and experience with Professor Bird. Inspiring!